What is the oxidation number of manganese in Mn so4 2?
 Olivia Owen
Olivia Owen | PubChem CID: | 24580 |
|---|---|
| Description: | Manganese(II) sulfate is a metal sulfate in which the metal component is manganese in the +2 oxidation state. It has a role as a nutraceutical. It is a metal sulfate and a manganese molecular entity. It contains a manganese(2+). ChEBI |
Similarly, what is the oxidation number of Mn in MnSO4?
The oxidation number for MnSO4 is 2. EXPLANATION: SO4 oxidation number is (-2). So, Mn should have (+2).
Also, what is the oxidation number of Mn in mno4 2? As you said Oxygen has the oxidation state of -2 so if we add up all the oxygens they have a -8 oxidation state. As the oxidation state of the ion is equal to its charge, the Mn has to "balance" the -8 to get it up to -1. Therefore the oxidation state of Mn in Mno4- is +7.
Then, what is the oxidation state of manganese in MN?
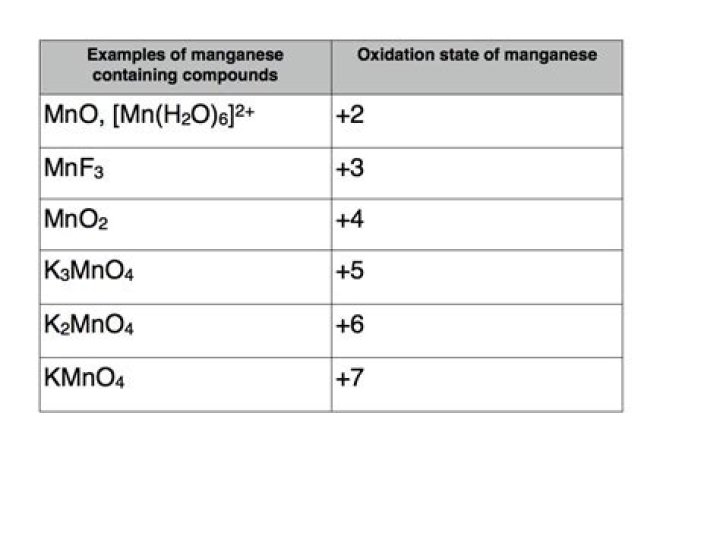
manganese has more than any other. In its compounds manganese exhibits oxidation states from +2 to +7. The common oxidation states are +2, +4, and +7, but the less common +3, +5, and +6 states are easily prepared.
What is the oxidation number of so4 2?
For example, in sulfate ion (SO42-), each oxygen has an oxidation number of -2, whereas sulfur has an oxidation number of +6.
Related Question Answers
What is the oxidation state of Mn in mncl2?
In MnCl2 , the oxidation state of Mn would be +2 .Which is the oxidation number of Sulphur in manganese sulphate?
The sulfate ion is SO42 -. The oxidation state of the sulfur is +6 (work it out!); therefore, the ion is more properly named the sulfate(VI) ion. The sulfite ion is SO32-.What is the oxidation number of k2cr2o7?
So, the total charge of potassium ions is +1(2)=+2, while oxygen ions is −2(7)=−14. Let x be the oxidation number of two chromiums. So, two chromium atoms have an oxidation number of +12.We need to find the oxidation number of Cr in K2Cr2O. 7.
| Related Questions & Answers | |
|---|---|
| What Is Matrix Theory | What Is Critical Angle |
What is the oxidation number of hno3?
Let's try an example. For nitric acid, HNO3, based on rule number 3 hydrogen has a +1 Oxidation Number and each oxygen atom has a -2 Oxidation Number. Since there are three oxygen atoms that is a total Oxidation Number of 3 x -2 = -6 from the oxygen atoms.What is the oxidation number of free elements?
The oxidation state of a free element (uncombined element) is zero. For a simple (monoatomic) ion, the oxidation state is equal to the net charge on the ion. For example, Cl– has an oxidation state of -1. When present in most compounds, hydrogen has an oxidation state of +1 and oxygen an oxidation state of −2.Which is the most stable oxidation state of Mn?
2+Why is Manganese not magnetic?
Pure manganese is not ferromagnetic because the distance between manganese atoms is too small. At this distance, the nearest neighbor moments have a lower energy when they point in opposite directions. (See: RKKY interaction .)Why does manganese have so many oxidation states?
Mn have the maximum number of unpaired electrons which are present in the d-subshell (5 electrons). Hence, Mn exhibits the high number of oxidation states which ranges from +2 to +7.Is manganese the same as magnesium?
Magnesium (Mg) and manganese (Mn) are essential plant nutrients. They are absorbed by plant roots as bivalent cations (Mg++ and Mn++). Deficiencies of both nutrients have been diagnosed in Michigan.| Nutrient | Sufficiency range (concentration in ppm) |
|---|---|
| Magnesium (Mg) | 1,600 to 6,000 |
| Manganese (Mn) | 20 to 50 |
Why is the two oxidation state of manganese more stable?
(1) It is seen from the electronic configuration of Mn2+ that d-orbital is half filled. (2) A half filled d-orbital is more stable as compared to empty or partially filled orbital. (3) Hence Mn2+ ion is more stable oxidation state of manganese.What is the oxidation number assigned to manganese in MnF4?
+4In which compound MN will not show +2 oxidation state?
Mn2+ is more stable due to half filled d-orbitals but Fe2+ is not stable because it does not have half filled d-orbitals. To what extent do the electronic configurations decide the stability of oxidation states in the first series of the transition elements? Illustrate your answer with example.Is manganese ore magnetic?
In manganese ores a series of complex oxides exist between hausmannite (Mn,O,) and jacob- site (FeMn),O, with varying magnetic properties. Of these, jacobsite is known to be very highly magnetic.Where is manganese found in the body?
Manganese is primarily found in the bones, liver, kidneys, and pancreas, according to the University of Maryland Medical Center, and helps the body form connective tissue, bones, blood-clotting factors, and sex hormones. According to Lenntech, manganese is essential not only to humans, but also to all species.What is the oxidation number of manganese in MnO4 1?
The oxidation number of Mn in [MnO4]− ion is +7.What is the oxidation state of Mn in MnO4 minus?
Sum of all the oxidation numbers in a polyatomic ion is equal to the charge on the ion. Thus, the total charge on the polyatomic anion is −1. Thus, the oxidation number of Mn in MnO−4 is +7.What is the oxidation state of Cl in cl2o?
as we know oxidation state of oxygen is -2 therefore the equation can be 2x+(-2)=0 where x is the oxidation state of chlorine ..now on solving we get answer as 1.Is MnO2 a peroxide?
Peroxides are compounds in which the oxidation state of Oxygen atoms is -1. Since you know that MnO2 is Mn(IV) oxide, which means Oxygen atoms are in -2 oxidation state. Hence it is not called a peroxide.Can oxidation number 0?
a) The net charge on neutral atoms or molecules is zero. So the overall oxidation state of them is zero. For example oxidation state of elemental atoms such as sodium, magnesium, iron is zero. The oxidation number of an atom in an oxygen molecule is zero.What is the name for MnO4 -?
Permanganate is a manganese oxoanion and a monovalent inorganic anion.What is the oxidation state of Mn in mn2o3?
+3What is the oxidation number of H in h2co3?
+1What is the oxidation number of Na?
+1Can so42 be oxidized?
Oxidation-Reduction:Sulfate is a very weak oxidizing agent. Since sulfur is in its maximum oxidation number in sulfate ion, this ion cannot act as a reducing agent.