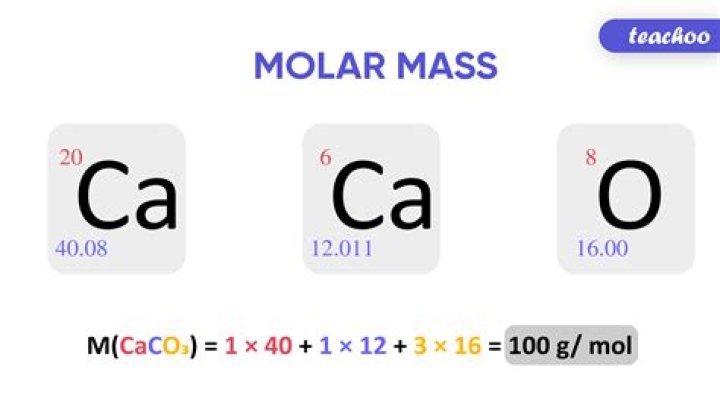
Cadmium sulfide / Molar mass 144.46 g/mol
In respect to this, what is the molar mass of CD?
112.411 u
Furthermore, what is the molar mass of CD no3 2? 236.42 g/mol
Herein, what is the molar mass of OS?
Osmocene
| Names |
| SMILES[show] |
| Properties |
| Chemical formula | C10H10Os |
| Molar mass | 320.42 g·mol−1 |
What is the mass in grams of a single atom of CD?
The mass of a single carbon atom is 1.994 x 10-23 g.
Related Question Answers
What is the correct name for CdS?
Cadmium sulfide
Is CdS soluble in water?
Physical and Chemical Properties of Cadmium CompoundsThe metal cadmium is insoluble in water (Weast 1985). Cadmium sulfide, CdS, has low solubility; its reported solubility limit is 1.3 mg/L (9.0 mol/ m3) at 20°C (Weast 1985).
What is CD in the periodic table?
Cadmium (Cd), chemical element, a metal of Group 12 (IIb, or zinc group) of the periodic table. Is CD a cation or anion?
Cadmium(2+) is a divalent metal
cation, a
cadmium cation and a monoatomic dication. It has a role as a cofactor.
4.3Related Element.
| Element Name | Cadmium |
| Atomic Number | 48 |
How many electrons does CD have?
48
Is cadmium a solid liquid or gas?
Phase of the elements
| Hydrogen | Gas | Strontium |
| Radon | Gas | Cadmium |
| Bromine | Liquid | Indium |
| Mercury | Liquid | Tin |
| Lithium | Solid | Antimony |
Is cadmium positive or negative?
The average concentration of
cadmium in Earth's crust is between 0.1 and 0.5 parts per million (ppm). It was discovered in 1817 simultaneously by Stromeyer and Hermann, both in Germany, as an impurity in zinc carbonate.
| Cadmium |
| Oxidation states | −2, +1, +2 (a mildly basic oxide) |
| Electronegativity | Pauling scale: 1.69 |
What is the atomic mass of chromium?
51.9961 u
What is the formula to find molar mass?
The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol). The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms. How many kg are in a mol?
0.001 Kilogram
What is formula unit of mass?
The mass of the formula unit of a substance is the sum of the atomic masses of all atoms in an ionic compound's formula unit. The trick to measuring an ionic compound's formula mass is to accurately count each atom in the formula and multiply its atoms' atomic masses accordingly. Is atomic mass equal to molar mass?
Molar mass is the mass of one mole of a substance. Atomic mass is the mass of one individual unit of a substance. the atomic mass in amu of a substance is numerically equivalent to the mass in g of one mole of that substance. What is mole in chemistry?
Mole, also spelled mol, in chemistry, a standard scientific unit for measuring large quantities of very small entities such as atoms, molecules, or other specified particles. What is molar mass simple?
Molar mass is defined as the mass in grams of one mole of a substance. The units of molar mass are grams per mole, abbreviated as g/mol. The mass of a single isotope of any given element (the isotopic atomic mass) is a value relating the mass of that isotope to the mass of the isotope carbon-12 ( How is mol calculated?
Use the molecular formula to find the molar mass; to obtain the number of moles, divide the mass of compound by the molar mass of the compound expressed in grams. How do we calculate atomic mass?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom. What is molar mass used for?
Molar mass is a ratio that is used to convert a mass measurement to an amount of substance. This amount is expressed as a number of particles, such as atoms, molecules or ions. It is the ratio between the mass of something and the number of particles that form it. What is the name for CrCl6?
Chromium Hexachloride
What is the formula for cadmium nitrite?
Cd(NO3)2
Is CD no3 2 soluble in water?
Cadmium nitrate
| Names |
| Boiling point | 132 °C (270 °F; 405 K) at 760 mmHg (tetrahydrate) |
| Solubility in water | 109.7 g/100 mL (0 °C) 126.6 g/100 mL (18 °C) 139.8 g/100 mL (30 °C) 320.9 g/100 mL (59.5 °C) |
| Solubility | Soluble in acids, ammonia, alcohols, ether, acetone 5 g/L in methanol |
What is the molar mass of Naoh?
39.997 g/mol
What is the name of CD no3 2?
Cadmium nitrate
What is the molar mass of na2s?
78.0452 g/mol
Is cadmium a transition metal?
Cadmium, a transition metal, has the chemical symbol of Cd. Cadmium is located in the d-block and 12 group of the periodic table possessing an atomic number of 48 and an atomic mass of 112.411g. Cadmium was discovered in Germany in 1817, by a German scientist, Friedrich Strohmeyer. Is cadmium chloride soluble?
Cadmium chloride is a white crystalline solid. It is soluble in water. What is the mass in grams of one silver atom?
So one silver atom weighs 1.79 x 10-22 g. How many atoms are in a mole?
The value of the mole is equal to the number of atoms in exactly 12 grams of pure carbon-12. 12.00 g C-12 = 1 mol C-12 atoms = 6.022 × 1023 atoms • The number of particles in 1 mole is called Avogadro's Number (6.0221421 x 1023). What is the mass of one atom?
Atomic mass, the quantity of matter contained in an atom of an element. It is expressed as a multiple of one-twelfth the mass of the carbon-12 atom, 1.992646547 × 10−23 gram, which is assigned an atomic mass of 12 units. In this scale 1 atomic mass unit (amu) corresponds to 1.660539040 × 10−24 gram. What is the mass in grams of one 12c atom?
Atomic mass: the mass in grams of one atom (1.992646 x 10-23 g for 12C). Atomic mass is different than the mass number A (#protons). How do you find mass in grams?
The molar mass is a physical property defined as the mass of a given substance (chemical element or chemical compound) divided by the amount of substance. The molar mass of atoms of an element is given by the standard relative atomic mass of the element multiplied by the molar mass constant, 1 × 10−3 kg/mol = 1 g/mol. What is the average mass in grams of one Zn atom?
The Elements, sorted by Atomic Mass
| Atomic Number | Symbol | Atomic Weight (amu, g/mol) |
| 28 | Ni | 58.70 |
| 27 | Co | 58.9332 |
| 29 | Cu | 63.546 |
| 30 | Zn | 65.38 |
 Michael Henderson
Michael Henderson