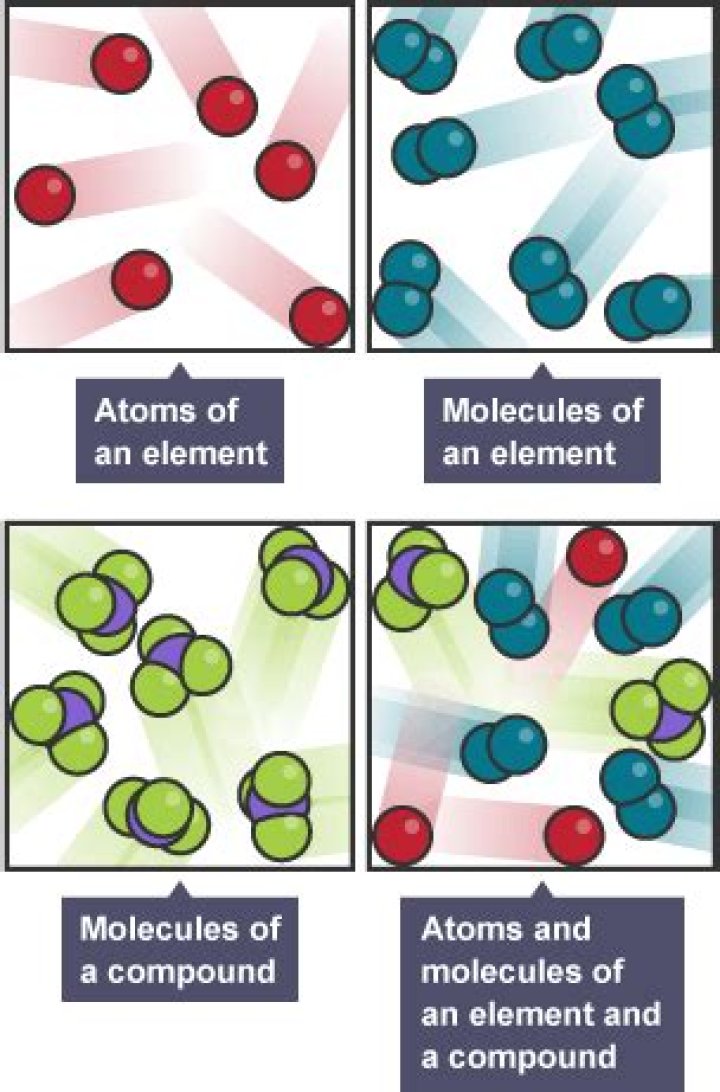
What is the chemical formula for the ionic compound strontium and bromine?
 David Schmidt
David Schmidt Also, what is the ionic formula for strontium bromide?
Strontium bromide (SrBr2)
Likewise, will sulfur and strontium form an ionic compound? The formula unit for the ionic compound made of strontium and sulfur is SrS, called strontium sulfide. Strontium, has a charge of 2+. It's an ionic compound, so sulfur - the non metal -'s ending would change to ide.
Hereof, does magnesium and bromine form an ionic compound?
For example, magnesium and bromine form an ionic bond. Magnesium has an oxidation number of +2 and bromine has an oxidation number of -1. The following shows an electron dot formula for the ionic compound that is formed by magnesium and bromine.
What are the formulas for ionic compounds?
Examples of Ionic Compounds
| Compound Name | Formula | Cation |
|---|---|---|
| sodium chloride | NaCl | Na+ |
| calcium chloride | CaCl2 | Ca2+ |
| iron(II) oxide | FeO | Fe2+ |
| aluminum sulfide | Al2S3 | Al3+ |
Related Question Answers
Is CBr4 ionic?
CBr4 and AlBr3 are both not ionic compounds. When an ionic compound precipitates, the ions become trapped in a crystal lattice.Is CaBr2 an ionic compound?
This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central.Ionic Compounds, Naming and Formula Writing.
| A | B |
|---|---|
| Calcium Bromide | CaBr2 |
| Calcium Iodide | CaI2 |
| Barium Fluoride | BaF2 |
| Barium Chloride | BaCl2 |
Is cs2 atomic molecular or ionic?
Together, they form an ionic compound and therefore, an ionic solid. b. CS2(s) - both are nonmetals which form a covalent compound. Therefore CS2 is a molecular solid.Does strontium and bromine form an ionic compound?
Explanation: It has two valence electrons and commonly forms Sr2+ ion. Bromine is a non-metal with seven valence electrons, and commonly forms the Br− ion. When strontium and bromine make music together, we conceive of a redox processDoes strontium bromide have a high melting point?
643 °CIs c6h12o6 ionic or covalent?
Sucrose (table sugar), C12H22O11 is MOLECULAR or COVALENT compound, while sodium chloride (table salt) is _ an IONIC compound. 6. Carbon monoxide, CO, is an example of a diatomic molecule, while ammonia and glucose, NH3 and C6H12O6, are examples of POLYATOMIC molecules.What is the formula for strontium phosphate?
Answer and Explanation: The formula for strontium phosphate is Sr3 (PO4)2. When strontium loses its two valence electrons it becomes Sr+2. The phosphate polyatomic ion is POWhat is the name for k2s?
potassium sulfideIs MgBr2 acid or base?
Therefore MgBr2 is a strong electrolyte. solubility rules it is soluble, and so it is a strong electrolyte. D. Ca(OH)2 is both a strong base, and an ionic compound.What is the chemical formula for the compound formed between magnesium and bromine?
Therefore, for the resulting compound to be neutral, two bromine anions must combine with one magnesium cation to form magnesium bromide (MgBr2).Is MgBr ionic?
Grignard reagents such as CH3MgBr are best thought of as hybrids of ionic and covalent Lewis structures. Grignard reagents are our first source of carbanions (literally, "anions of carbon").What is an ionic formula?
The overall ionic formula for a compound must be electrically neutral, meaning it has no charge. When writing the formula for the ionic compound, the cation comes first, followed by the anion, both with numeric subscripts to indicate the number of atoms of each.Is co2 an ionic compound?
Answer and Explanation: CO2 is a molecular compound. Ionic compounds are composed of a non-metal and a metal element. Molecular compounds are made up of two non-metals.Is magnesium bromide a metal or nonmetal?
It is neither a metal nor a nonmetal. Magnesium bromide, MgBr2 , is an ionic compound containing the metal magnesium (Mg) and the nonmetal bromine (Br).What ionic compound is formed by the ions al3+ and o2?
"Names and Formulas of Inorganic Compounds"| A | B |
|---|---|
| C4- | carbide ion |
| formula of Al3+ and O2- | Al2O3 |
| formula of Al3+ and S2- | Al2S3 |
| formula of Al3+ and Cl- | AlCl3 |
Do ionic bonds form molecules?
When atoms join together, they form either molecules or ionic compounds, depending on whether they are joined by covalent bonds or ionic bonds. When atoms join together by covalent bonds, they form “molecules”. When atoms join together by ionic bonds, they form “ionic compounds”.Which compound contains ionic bonds?
Ionic bond examples include:- LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.
Can two metals form an ionic compound?
An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. A covalent bond involves a pair of electrons being shared between atoms.What is CU 2s?
Copper(I) sulfide is a copper sulfide, a chemical compound of copper and sulfur. It has the chemical compound Cu2S. It is found in nature as the mineral chalcocite.What is the formula for the ionic compound ammonium chloride?
Ammonium chloride | NH4Cl - PubChem.What is the chemical name for Cu2S?
copper(I) sulfideWhat is the name of the ionic compound Cu3P?
Binary Ionic Compounds| A | B |
|---|---|
| copper (I) oxide | Cu2O |
| copper (I) sulfide | Cu2S |
| copper (I) nitride | Cu3N |
| copper (I) phosphide | Cu3P |