What are reactants and products in a chemical equation?
 Michael Henderson
Michael Henderson Also question is, what are the products in a chemical equation?
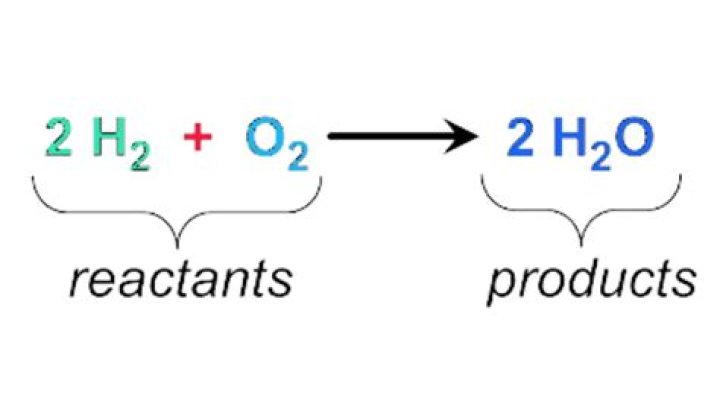
The substance(s) to the left of the arrow in a chemical equation are called reactants. A reactant is a substance that is present at the start of a chemical reaction. The substance(s) to the right of the arrow are called products . A product is a substance that is present at the end of a chemical reaction.
Also, what do reactants form during chemical reactions? In a chemical reaction, the atoms and molecules that interact with each other are called reactants. No new atoms are created, and no atoms are destroyed. In a chemical reaction, reactants contact each other, bonds between atoms in the reactants are broken, and atoms rearrange and form new bonds to make the products.
Similarly, it is asked, what are the examples of reactants and products?
In the burning of natural gas, for example, methane (CH4) and oxygen (O2) are the reactants in the chemical reaction. are the substances formed by a chemical reaction. In the burning of natural gas, carbon dioxide (CO2) and water (H2O) are the products formed by the reaction.
What does a chemical equation do?
A chemical equation is the symbolic representation of a chemical reaction in the form of symbols and formulae, wherein the reactant entities are given on the left-hand side and the product entities on the right-hand side.
Related Question Answers
How do you write reactants and products?
In a chemical equation, the reactants are written on the left, and the products are written on the right. The coefficients next to the symbols of entities indicate the number of moles of a substance produced or used in the chemical reaction.What's the easiest way to balance a chemical equation?
There is a strategy that will help you balance equations more quickly. It is called balancing by inspection. Basically, you look at how many atoms you have on each side of the equation and add coefficients to the molecules to balance out the number of atoms.What are examples of products?
A product can be physical or virtual. Physical products include durable goods (such as cars, furniture, and computers) and nondurable goods (such as food and beverages). Virtual products are offerings of services or experiences (such as education and software).What are reactants and products?
Chemical reactions occur when chemical bonds between atoms are formed or broken. The substances that go into a chemical reaction are called the reactants, and the substances produced at the end of the reaction are known as the products.What do products mean?
Definition: A product is the item offered for sale. A product can be a service or an item. It can be physical or in virtual or cyber form. Every product is made at a cost and each is sold at a price. The price that can be charged depends on the market, the quality, the marketing and the segment that is targeted.Is oxygen a reactant or product in photosynthesis?
In photosynthesis, oxygen, carbon dioxide, ATP, and NADPH are reactants. GA3P and water are products. In photosynthesis, chlorophyll, water, and carbon dioxide are reactants. GA3P and oxygen are products.What happens during a chemical reaction?
Chemical reaction, a process in which one or more substances, the reactants, are converted to one or more different substances, the products. Substances are either chemical elements or compounds. A chemical reaction rearranges the constituent atoms of the reactants to create different substances as products.What is a products in chemistry?
Products are the species formed from chemical reactions. During a chemical reaction reactants are transformed into products after passing through a high energy transition state.Is glucose a reactant or product?
Glucose is the reactant and alcohol and carbon dioxide are the products.What does the arrow represent in a chemical equation?
Reaction arrows are used to describe the state or progress of a reaction. The chemical reaction arrow is one straight arrow pointing from reactant(s) to product(s) and by-products, sometimes along with side products.What are the two types of chemical reactions?
Representation of four basic chemical reactions types: synthesis, decomposition, single replacement and double replacement.What two types of chemical equations are there?
The Common Types of Chemical Reactions- Combination.
- Decomposition.
- Single displacement.
- Double displacement.
- Combustion.
- Redox.





