Is n2o4 polar or nonpolar?
 Olivia Owen
Olivia Owen Then, what type of bond is n2o4?
covalent bonds
Beside above, does n2o have a polar bond? If you look at the Lewis structure for N2O it appears to be a symmetrical molecule. However, to determine if N2O is polar we consider the molecular geometry or shape of the molecule. Polarity results from an unequal sharing of valence electrons. Therefore N2O is a polar molecule.
Hereof, is dinitrogen polar or nonpolar?
It has zero dipole moment. Two N atoms in nitrogen molecule have zero electronegativity difference. The bond pairs of electrons are equally distributed between two N atoms.
How do you know if its polar or nonpolar?
Step 2: Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.
Related Question Answers
Does k2o covalent bond?
K2O is ionic. Ionic bonds are formed between metals and non-metals, while covalent bonds are formed between non-metals only. Therefore K2O is ionic.What type of bond is pcl3?
Hybridization of PCl3 (Phosphorus Trichloride)| Name of the Molecule | Phosphorus Trichloride |
|---|---|
| Molecular Formula | PCl3 |
| Hybridization Type | sp3 |
| Bond Angle | Less than 109o |
| Geometry | Trigonal Pyramidal |
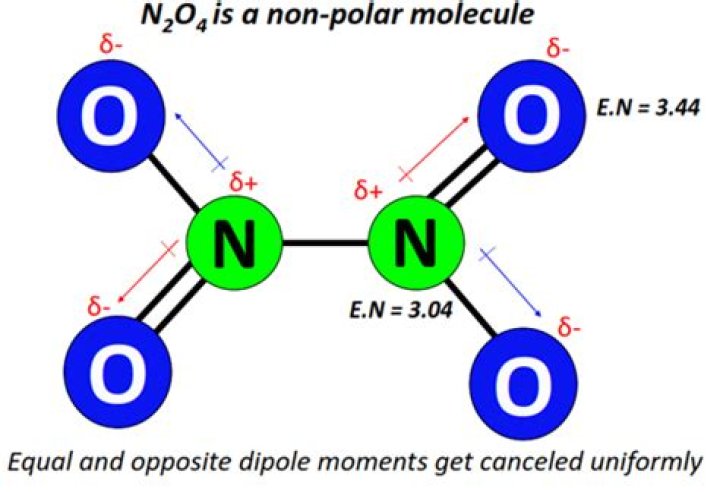
What is the Lewis structure of n2o4?
In lewis structure of N2O4 ion, there are two double bonds (N=O) between oxygen and nitrogen, two N-O single bonds and one N-N bond. In two oxygen atoms, each have three lone pairs with a -1 charge. Like these lone pairs can be transferred as bonds.Does n2o5 have coordinate bond?
From the structure, it is clear that each nitrogen is bonded to three oxygen atoms. One oxygen atom is doubly bonded to the nitrogen; the other forms a sigma bond with nitrogen and the third oxygen atom forms a coordinate covalent bond with nitrogen. Thus, N2O5 have both covalent and coordinate covalent bonds.Is MgH2 ionic or covalent?
Yes, Magnesium hydride (MgH2) is an ionic compound.What type of bond is AlF3?
ionic bondingIs bacl2 ionic or covalent?
Explanation: Barium chloride is an ionic compound composed of one barium cation and two chlorine anions. In this case, chlorine will want to have a −1 oxidation state due to its high electronegativity.How do you calculate the bond order of n2o4?
Hey saini first of all find the no of charge on n2o4 which is minus then find no of charge delocalised on how many atom which is 1/4 to every oxygen and then sum the delocalised pie bondto one where 1is being added for sigma character which is always 100% so 1/2 +1=1.5or another method the easiest method draw theIs CH2Cl2 polar or nonpolar molecule?
CH2Cl2 polar molecule The molecule is tetrahedral due to 4 electron repulsions around the central C atom. Although the bond arrangement around the C atom in CH2Cl2 is symmetrical, the differing polarities of the C–H and C–Cl bonds means the effect of the polar bonds is not cancelled, so the molecule is polar.Is CCl4 a polar or nonpolar molecule?
If you look at the Lewis Structure for CCl4 it appears to be a symmetrical molecule. While there may be unequal sharing of electrons in the individual bonds, in a nonpolar molecule like CCl4 these bonds are evenly distributed and cancel out. There is no net dipole and the CCl4 is non-polar.Is n2 polar and why?
A polar molecule has a net electric dipole moment. Since N2 is a symmetric molecule without a net electric dipole moment, N2 is not polar. A nitrogen atom has five electrons in its outermost electron shell. Hence, two nitrogen atoms bond with each other by sharing three electrons with each other.Is ammonia polar or nonpolar?
Ammonia is a polar molecule:The electrostatic potential clearly shows that the nitrogen is partially negative whereas the hydrogens are partially positive.
Is water polar or nonpolar?
Water is a polar molecule. While the overall charge of the molecule is neutral, the orientation of the two positively charged hydrogens (+1 each) at one end and the negatively charged oxygen (-2) at the other end give it two poles.Is nitrogen and oxygen polar or nonpolar?
Bonds between carbon and other elements such as oxygen and nitrogen are polar. The polarity of a bond depends on the electronegativities of the bonded atoms. Large differences between the electronegativities of the bonded atoms increase the polarity of bonds.Which is more polar no2 or n2o?
Polarity arises from a difference in electronegativity. If we consider NO2 and N2O both, there is some electronegativity difference. So, both are polar. As we know that NO2 is bent structure whereas N2O is linear, so the dipole moment is more in NO2 and hence it is more polar.Is PCl3 polar or nonpolar?
PCl3 is polar, because of its trigonal pyramidal structure. Only 3 of the 5 valence electrons of the phosphorus are used for bonding with chlorine, so the other two are unshared.Is CH3Cl polar or nonpolar?
Since the H is between B and C in terms on electronegativity values, their difference in electronegativity values is so small, the C-H bond is considered nonpolar; thus, no dipole arrow is drawn for the C-H bonds. Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.Is ozone a polar or nonpolar bond?
In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element).What is polar and nonpolar?
Nonpolar bonds form between two atoms that share their electrons equally. Polar bonds form when two bonded atoms share electrons unequally.What is the difference between a polar and nonpolar solvent?
Polar solvents have large dipole moments (aka “partial charges”); they contain bonds between atoms with very different electronegativities, such as oxygen and hydrogen. Non polar solvents contain bonds between atoms with similar electronegativities, such as carbon and hydrogen (think hydrocarbons, such as gasoline).What is the difference between a polar and nonpolar covalent bond?
A covalent bond that has an unequal sharing of electrons, as in part (b) of Figure 4.4. 1, is called a polar covalent bond. A covalent bond that has an equal sharing of electrons (part (a) of Figure 4.4. 1) is called a nonpolar covalent bond.Which Vsepr shapes are polar and nonpolar?
Lewis Structures and the Shapes of Molecules| Formula | 3D Structure Shape Polarity | |
|---|---|---|
| 1. | CH4 | tetrahedral nonpolar |
| 2. | NH3 | trigonal pyramidal polar |
| 3. | H2O | bent polar |
| 4. | H3O+ | trigonal pyramidal charged |
Is tetrahedral polar or nonpolar?
Any 100% symmetrical tetrahedral molecule will be nonpolar.Tetrahedral molecules have no nonbonding electron pairs and all identical bond angles. Therefore, the only way they can be asymmetric is if one atom is different from the rest.





