Liquid acetic acid is a hydrophilic(polar) protic solvent, similar to ethanol and water. With amoderate relative static permittivity (dielectric constant) of 6.2,it dissolves not only polar compounds such as inorganicsalts and sugars, but also non-polar compounds such as oilsas well as polar solutes. In this regard, is acetic acid polar?
Since it is a double bond, the electrons on the oxygenmake acetic acid extremely polar unless there existexcessive dipole cancellations in the same framework, which usuallydoesn't happen. In simple terms, it is a polar molecule,which y means that there is one positive and one negative end tothe same.
Also, what type of bond is acetic acid? The chemical structure of carboxylic acid showsthat the first carbon on the left has single bonds withthree hydrogen atoms. The second carbon, on the other hand, has adouble bond with the oxygen atom and is singly bonded withthe first carbon and the -OH group.
Keeping this in consideration, is acetic acid polar covalent?
Polarity. Acetic acid is a polar molecule.Meaning it has a positive end and a negative end. This is becausethe oxygen atoms have unshared electron pairs.
Is acetic acid hydrophilic?
Acetic acid is a colourless liquid organiccompound with the chemical formula CH3COOH (also written as CH3CO2Hor C2H4O2). When undiluted, it is sometimes called glacialacetic acid.Liquid acetic acid is ahydrophilic (polar) protic solvent, similar to ethanol andwater.
Related Question Answers
Is water Polar?
A water molecule, because of its shape, is apolar molecule. That is, it has one side that is positivelycharged and one side that is negatively charged. The molecule ismade up of two hydrogen atoms and one oxygen atom. The bondsbetween the atoms are called covalent bonds, because the atomsshare electrons. Who discovered acetic acid?
The first person to extract acetic acid fromvinegar was the Muslim alchemist Jabir ibn Hayyan Geber (c.721–815). The pure compound was not produced, however, foranother ten centuries when the German chemist Georg Ernst Stahl(1660–1734) extracted acetic acid from vinegar in 1700by distillation. What bond in acetic acid is most polar?
Answer: (-C=O) bond in the acetic acid isthe most most polar bond. Explanation: Acetic acid isan organic acid with carboxyl group (-COOH). In thisfunctional group two oxygen atoms are present in which one oxygenatom is double bonded to carbon atom and another one is singlebonded to the carbon atom. Is ch3cooh a vinegar?
CH3COOH is an accepted and common form of writingthe structure of acetic acid (commonly known as vinegar whendiluted in water). Why acetic acid is soluble in water?
The presence of this small amount of ions results inaqueous acetic acid being a weak electrolyte. Why isacetic acid highly soluble in water? Water hasa network of hydrogen bonds between molecules in its liquid phaseand so when a substance dissolves in water this bonding isdisrupted. Is Vinegar an oxidizer?
Vinegar is an acid (in fact, it's known as aceticacid). The hypochlorous acid then reacts with the rest of thevinegar, Dr. Biberdorf continues. Because hypochlorous acidis something that's called an oxidizing agent, it takeselectrons from whatever it comes into contact with. Is acetic acid harmful?
Acetic Acid Hazards. Acetic acid can be ahazardous chemical if not used in a safe andappropriate manner. This liquid is highly corrosive to the skin andeyes and, because of this, must be handled with extreme care.Acetic acid can also be damaging to the internalorgans if ingested or in the case of vapor inhalation. What contains acetic acid?
Ingredient Results - Acetic Acid. A naturallyoccurring acid found in a variety of plants and fruits suchas apples, grapes, oranges, pineapples, and strawberries. It is anorganic acid that gives vinegar its sour taste and distinctsmell. It Is produced through a fermentation process. Is co2 polar or nonpolar?
Is Carbon Dioxide (CO2) Polar OrNonpolar? Carbon dioxide (CO2) is nonpolarbecause it has a linear, symmetrical structure, with 2 oxygen atomsof equal electronegativity pulling the electron density fromcarbon at an angle of 180 degrees from eitherdirection. Is methanol polar or nonpolar?
Methanol is a polar molecule: See graphic on the left. The alcohol (-OH) groupdominates the molecule making it definitely polar. Theelectrostatic potential clearly shows that the oxygen is partiallynegative whereas the carbon and hydrogens are partiallypositive. Is caffeine a polar molecule?
The hydrogen of the water molecule is attractedto the highly electronegative nitrogen and oxygen atoms.Caffeine is a polar molecule due to theelectronegativity difference between the carbon-oxygen singlepolar covalent bonds and carbon nitrogen single polarcovalent bonds. Is olive oil polar or nonpolar?
Most nonpolar compounds do not dissolve inpolar compounds. In other words, they are insoluble inpolar compounds. For example, olive oil is a mixtureof nonpolar compounds. Olive oil does not dissolve inwater. Why is dichloromethane polar?
The individual bonds within dichloromethane aresomewhat polar due to the electronegativity differencesbetween C&H and between C&Cl, so yes, it is polar atthe bond level. However, the net polarity is fairly small, so thatthe molecule is barely polar. Is water polar nonpolar or ionic?
Water is a polar molecule because itsoxygen is strongly electronegative and, as such, pulls the electronpair towards itself (away from the two hydrogen atoms), thusacquiring a slightly negative charge. Is aspirin a polar?
Aspirin contains polar functional groupswhich can form hydrogen bonds with polar water molecules.Aspirin is more soluble in basic (alkaline) solutions, so itreadily dissolves in the duodenum which is the first part of theintestine. What is the Colour of acetic acid?
Acetic acid, also known as ethanoic acid,is an organic chemical compound best recognized for giving vinegarits sour taste and pungent smell. Pure water-free aceticacid (glacial acetic acid) is a colorless hygroscopicliquid and freezes below 16.7 °C (62 °F) to a colourlesscrystalline solid. What is the common name of acetic acid?
The common name for ethanoic acid is'acetic acid.' A solution of 4 percent or more aceticacid and water is called 'vinegar.' The formula for aceticacid is CH3COOH. How Acetic acid is manufactured?
Acetic acid is produced industrially bothsynthetically and by bacterial fermentation. About 75% of aceticacid made for use in the chemical industry is made bythe carbonylation of methanol, explained below. Can acetic acid be stored in plastic?
Because of its low conbustion temperature, glacialacetic acid is best stored in a refrigerator or -4degree Centigrade freezer. It should not be stored nearstrong oxidizers like sodium peroxide, nitric acid andchromic acid, strong acids, or strong bases likechlorine bleach. Is acetic acid strong or weak?
A weak acid is an acid that ionizes onlyslightly in an aqueous solution. Acetic acid (found invinegar) is a very common weak acid. Is there hydrogen bonding in acetic acid?
Acetic acid (the ninth entry) is an interestingcase. A dimeric species, shown on the right, held together by twohydrogen bonds is a major component of the liquidstate. Why is acetic acid weak?
Weak acids partially ionize in water. This meansthat only SOME of the acid molecules break up into positiveand negative ions when a weak acid is dissolved in water.Example: Acetic acid ( ) is a weak acid because onlysome of the molecules break up into and ions when dissolved inwater. How do you know a molecule is polar?
Step 2: Identify each bond as either polaror nonpolar. (If the difference in electronegativity for theatoms in a bond is greater than 0.4, we consider the bondpolar. If the difference in electronegativity is lessthan 0.4, the bond is essentially nonpolar.) If there are nopolar bonds, the molecule is nonpolar. What is acetic acid solution?
Acetic acid is an antibiotic that treatsinfections caused by bacteria or fungus. Acetic acid otic(for the ear) is used to treat infections in the ear canal. Thismedicine will not treat an inner ear infection (also called otitismedia). Acetic acid may also be used for purposes not listedin this medication guide. What is vinegar made of?
Vinegar is made from a variety of dilutedalcohol products, the most common being wine, beer, and rice.Balsamic vinegar is made from the Trebbiano andLambrusco grapes of Italy's Emilia-Romagna region. Some distilledvinegars are made from wood products such asbeech. Why is it called glacial acetic acid?
Acetic acid that contains a very low amount ofwater (less than 1%) is called anhydrous (water-free)acetic acid or glacial acetic acid. The reason it'scalled glacial is because it solidifies into solid aceticacid crystals just cooler than room temperature at 16.7 °C,which ice. 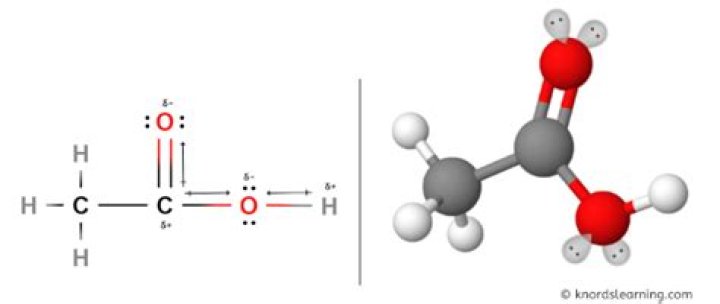
 John Peck
John Peck 




