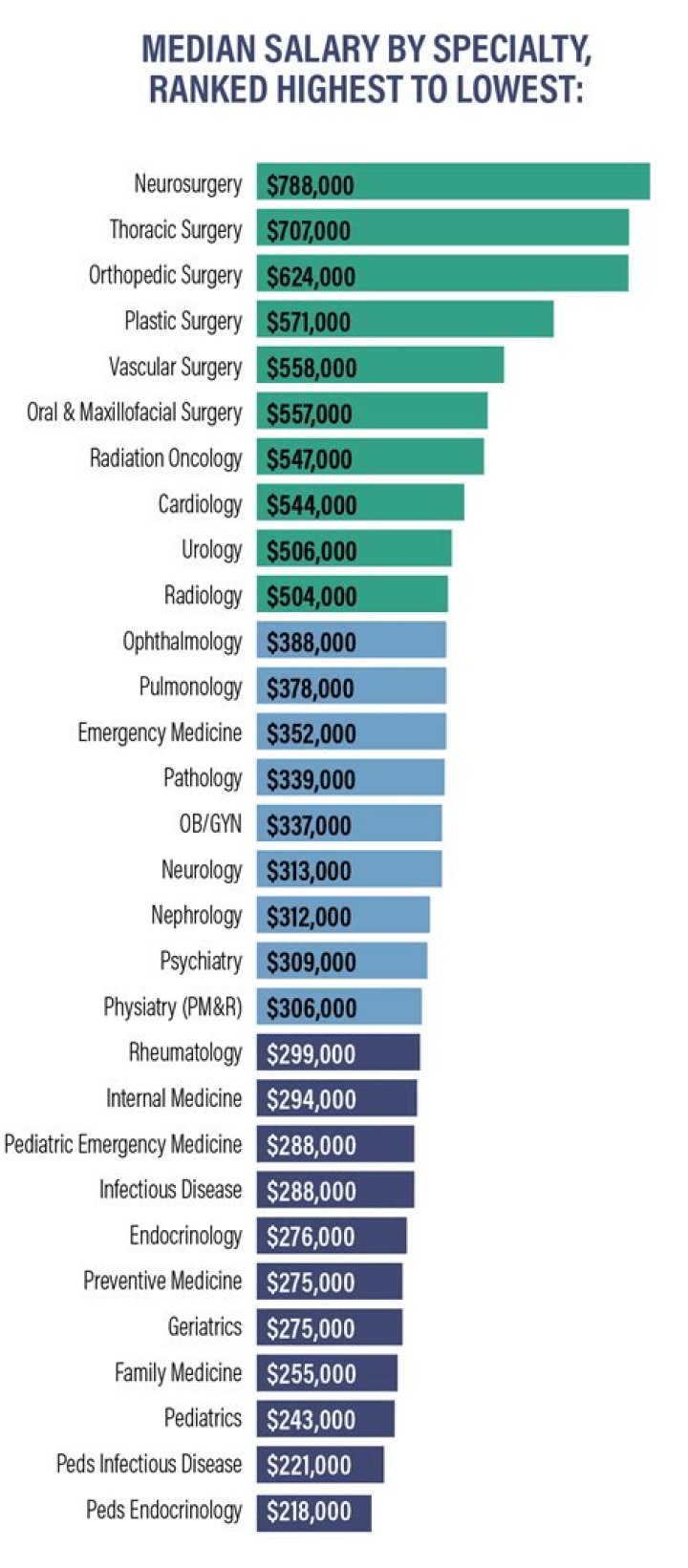
In the case of zinc and aluminum, there is only a slight chance of a reaction because of the relatively small change in potential between the two metals and the formation of an insulating film on the surface of the aluminum. One of the key factors in the reaction between dissimilar metals is the contact surface area. Considering this, does Zinc react with aluminum?
Galvanized steel screws, however, are plated with a corrosion-resistant coating, usually consisting of zinc, that is not nearly as reactive with aluminum. The zinc plating prevents the underlying steel from coming into contact with the aluminum, and the risk of corrosion of the aluminum is reduced significantly.
Similarly, what metals should not be used together? Metals farther apart should not be used together. For example, bronze and copper can be used together; aluminum and copper should not.
Just so, what metal is compatible with aluminum?
Coated Steel With a thick enough coating, even a very reactive metal like brass can be used on an aluminum structure without corrosion. Since stainless steel remains one of the least reactive metals without coating, it's smarter to use it as the base material.
What metals react with zinc?
For example, zinc metal reacts with hydrochloric acid producing zinc chloride and hydrogen gas. Bases also react with certain metals like zinc or aluminum for example to produce hydrogen gas. For exanple, sodium hydroxide reacts with zinc and water to form sodium zincate and hydrogen gas.
Related Question Answers
Is Zinc stronger than aluminum?
Zinc alloys are some of the strongest and toughest materials for die casting. Zinc has electrical conductivity than aluminum, and better conductor of heat than Aluminum which makes it perfect for applications like heat sinks or electrical components. Can you mix galvanized and aluminum?
Under atmospheric conditions of moderate to mild humidity, contact between a galvanized surface and aluminum or stainless steel is unlikely to cause substantial incremental corrosion. However, under very humid conditions, the galvanized surface may require electrical isolation from the aluminum or stainless steel. What is the difference between aluminum and zinc?
The main difference between the two are their weight and melting point. Aluminium is lighter than zinc, but zinc has a lower melting point. Another important difference is that casting precision is significantly better and the strength of zinc is considerably higher, so wall thickness can be reduced. Will galvanized bolts react with aluminum?
Under atmospheric conditions of moderate to mild humidity, contact between a galvanized surface and aluminum or stainless steel is unlikely to cause substantial incremental corrosion. However, under very humid conditions, the galvanized surface may require electrical isolation from the aluminum or stainless steel. How do you fasten aluminum?
Self-tapping screws, also called sheet metal screws, are very versatile. They can be used to securely fasten metals together, including aluminum. They can also be used to efficiently fasten metal to other materials, such as wood or plastic, by creating a much stronger bond than a regular nail. What metal does not react with aluminum?
Coated Steel With a thick enough coating, even a very reactive metal like brass can be used on an aluminum structure without corrosion. Since stainless steel remains one of the least reactive metals without coating, it's smarter to use it as the base material. Is Zinc compatible with stainless steel?
The stainless steels, including 304 and 316, are more positive than zinc and steel, so when stainless steel is in contact with galvanised steel and is wet, the zinc will corrode first, followed by the steel, while the stainless steel will be protected by this galvanic activity and will not corrode. Can you use stainless screws in aluminum?
When assembling aluminum panels, you want to use the strongest fasteners possible to ensure that your project holds together in high winds and winter weather. Despite the dissimilar metals and risk of corrosion, stainless steel screws are the recommended fastener for aluminum panels. What fasteners to use with aluminum?
Stainless Steel Screws As is the case with carbon steel screws, a plated stainless steel screw is less likely to corrode aluminum; screws treated with a high-quality coating consisting of zinc and aluminum flakes are especially resistant to corrosion. What happens if you mix aluminum and copper?
Copper and aluminum can be combined to form a copper-aluminum alloy. However, under very high temperature, copper and aluminum can form a solid solution. When this solution cools, the intermetallic compound CuAl2, or copper aluminide, can form as a precipitate. Is stainless steel and aluminum compatible?
Stainless steel fasteners in aluminium plates or sheets are normally considered safe, whereas aluminium rivets or bolts holding stainless steel parts together is an unwise combination, as there is a practical risk of corrosion. Even with no insulation between the metals, there should be little risk of corrosion. Can you use brass fittings in aluminum?
Brass is incompatible with aluminum, per Mil-STD-889 [affil. link to spec at Techstreet] (and Notices 1, 2 & 3 thereof) DISSIMILAR METALS. Galvanized is your best choice. Can steel and aluminum touch?
“Note that aluminum is close in activity with steel. However, they are still dissimilar metals and are subject to considerable galvanic corrosion when in contact in a corrosive atmosphere. If this condition exists, the smaller active metal could corrode at an accelerated rate. Can I use brass fittings on aluminum?
As the series suggests, steel and aluminum are relatively compatible, but if brass and steel contact, the steel will corrode because it is more anodic than the brass. The aluminum will also corrode where its exposed surface contacts the brass plate because brass is more cathodic. Can you put aluminum and steel together?
This is called galvanic corrosion. When you join aluminum and steel together, the aluminum will be preferentially corroded. Use an isolating coating or paint on the aluminum and the steel to isolate them electrically. However, all of your effort will go for nothing if you just bolt the joint together. Is it possible to weld steel to aluminum?
You can weld aluminum to most other metals relatively easily via adhesive bonding or mechanical fastening. However, in order to weld aluminum to steel, special techniques are required. To avoid this, you must isolate the other metal from the molten aluminum during the arc welding process. What do you put between aluminum and steel?
Put some rubber gasket material between them. 1/16 thk or use plastic from anti-freeze jugs anything to seperate the two metals. Zink or Stainless are the two worst to put next to alum. Is it OK to connect brass to stainless steel?
Connecting stainless steel plumbing pipes to brass fittings and pipes is risky. In the case of stainless steel (410) and brass, they are seven metals apart, which means they would be likely to corrode if connected directly. When it comes to stainless steel (301, 304 and 310) and brass, they are only two metals apart. Can you mix copper and galvanized?
Galvanized pipes should not be connected directly to copper pipes, period. The only time one can successfully replace a section of galvanized pipe with a section of copper is to make use of a dielectric union to ensure the metals do not have direct contact. Brass is an alloy of copper and nickel. Does copper react with aluminum?
A. Aluminum will be very susceptible to galvanic corrosion in contact with copper, assuming that the two metals are also in contact with a common electrolyte (such as water with some ionic content.) This will improve bolted joints by reducing the resistance, and resists corrosion. Does silver react with stainless steel?
At room temperature, they don't react with each other…so, if you keep, gold, silver and stainless steel together, in dry environment, they stay as they were… What makes stainless steel more corrosion resistant than iron? Does mild steel react with Aluminium?
The aluminium will corrode quickly, and so aluminium fasteners in stainless steel are not acceptable. However, a stainless screw in aluminium is frequently used although corrosion of the aluminium immediately around the stainless is quite possible. What metals can go together?
Match metals to your color palette. If your room has more warm colors, stick to warm-hued metals like copper and gold. If it's a cool palette, opt for cast iron or stainless steel. If your room has a neutral palette, such as white or black, you can be more liberal with how you mix and match different hues. What metal can touch copper?
It is not necessary to isolate copper from lead, tin or stainless steel under most circumstances. The principal metals of concern in terms of direct contact are aluminum and zinc. Iron and steel are generally not a problem unless their mass is similar or smaller than that of the copper. Is brass or stainless steel better for plumbing?
Brass is also able to absorb and withstand more heat than plastic, making brass valves an optimal choice for at home plumbing systems. One significant difference between stainless steel and brass valves is that brass valves tend to accept welding better than stainless steel valves. This offers some versatility. Why is zinc metal important?
It tarnishes in air. Most zinc is used to galvanise other metals, such as iron, to prevent rusting. Large quantities of zinc are used to produce die-castings, which are important in the automobile, electrical and hardware industries. Zinc is also used in alloys such as brass, nickel silver and aluminium solder. Is zinc a hard metal?
The metal is hard and brittle at most temperatures but becomes malleable between 100 and 150 °C. Above 210 °C, the metal becomes brittle again and can be pulverized by beating. Zinc is a fair conductor of electricity. How can you tell if a metal is zinc?
Zinc is naturally dull grey and is very hard to polish. Zinc has has an oxide that flakes off carrying some of the zinc so other things are coated in it so the zinc "rusts" instead of the base metal, this is called galvanization. What is another name for zinc?
Acétate de Zinc, Acexamate de Zinc, Aspartate de Zinc, Atomic Number 30, Chlorure de Zinc, Citrate de Zinc, Gluconate de Zinc, Méthionine de Zinc, Monométhionine de Zinc, Numéro Atomique 30, Orotate de Zinc, Oxyde de Zinc, Picolinate de Zinc, Pyrithione de Zinc, Sulfate de Zinc, Zinc Acetate, Zinc Acexamate, Zinc Is zinc a transition metal?
What is a transition metal? Not all d block elements count as transition metals! A transition metal is one that forms one or more stable ions which have incompletely filled d orbitals. On the basis of this definition, scandium and zinc do not count as transition metals - even though they are members of the d block. Why does zinc only form a 2+ ion?
Zinc has an electron configuration of [Ar] 3d10 4s2. On the fourth (and outermost) electron shell, Zinc has only two electrons before coming to a closed n=3 shell. Losing all the electrons on the fourth shell would mean Zn lost two negative charges, making it the ion Zn+2. What can zinc combine with?
Zinc is a fairly reactive metal that will combine with oxygen and other non-metals. Zinc metal tarnishes in moist air and burns in air to form the white zinc oxide. It is a fair electrical conductor and will react with dilute acids to release hydrogen. Zinc does not react with water. Does Zinc rust in water?
Like all ferrous metals, zinc corrodes when exposed to air and water. However, zinc corrodes at a rate of 1/30 of that for steel. Also like other ferrous metals, zinc corrodes or rusts at different rates depending on its environment (8). The patina layer is the products of zinc corrosion and rust. Why does Zinc react with hydrochloric acid?
Zn + HCl = H2 + ZnCl2 Zinc is oxidised (it becomes positive) and water is neutralized. It is a reduction-oxidation reaction where one of the reactants loses oxygen and the other one gains. So, answering your question, we get hydrogen gas and a salt. Hydrogen gas, is identified by exposing it to a burning flame. 
 Olivia Owen
Olivia Owen