Are elimination reactions endothermic?
 Joseph Russell
Joseph Russell Also asked, do elimination reactions need heat?
But at high temperatures, the Gibbs energy of activation ( ΔG‡ ) for elimination starts to be lower in energy than that for substitution reactions, and hence we get an increase in the amount of elimination product. Again, the bottom line is that, all else being equal, heat will tend to favor elimination reactions.
Also Know, are e2 reactions exothermic? Now with the E2 reactions, it happens to be that the more stable the product, the more stable the transition state, the faster the rate of reaction. What I am confused is the fact that E2 reactions are exothermic, and thus the transition state should resemble the reactants and not the products.
Similarly, you may ask, why do elimination reactions occur?
Elimination reactions are important as a method for the preparation of alkenes. The term "elimination" describes the fact that a small molecule is lost during the process. A 1,2-elimination indicates that the atoms that are lost come from adjacent C atoms.
What conditions favor elimination over substitution?
In the presence of a strong base, E2 will be the favored pathway. Elimination is typically preferred over substitution unless the reactant is a strong nucleophile, but weak base. Substitution is typically preferred over elimination unless a strong bulky base is used.
Related Question Answers
Is elimination exothermic or endothermic?
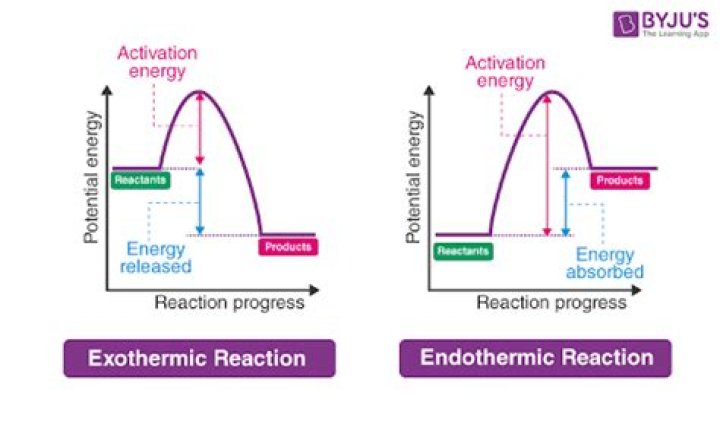
Why are elimination reactions endothermic? The reaction going from right to left is exothermic, so, perforce, the reaction left to right must be endothermic. The major factor is the bond energy of the carbon-carbon double bond is considerably less than two single C - C bonds.What does heat do in an organic reaction?
Generally speaking, heat will help speed up a chemical reaction, or drive a chemical reaction that wouldn't be able to occur otherwise.Is ch3oh sn1 or sn2?
The 2nd involving CH3OH as the nucleophile is SN1. Both involve a secondary alkyl halide which can be used in either SN2 or SN1 so this is of no help. The key is the nucleophiles.Does temperature affect sn2 reactions?
1 Answer. Truong-Son N. The higher the temperature, the faster a non-biological reaction tends to occur. For SN1 and SN2 reactions, the higher the temperature, the more elimination products you get.Is Solvolysis sn1 or e1?
Solvolysis is a type of nucleophilic substitution (SN1) /( SN2) or elimination, where the nucleophile is a solvent molecule. Characteristic of SN1 reactions, solvolysis of a chiral reactant affords the racemate.Why do organic reactions have high activation energies?
Activation energy and reaction rate Specifically, the higher the activation energy, the slower the chemical reaction will be. This is because molecules can only complete the reaction once they have reached the top of the activation energy barrier.How can you tell the difference between sn1 and sn2 reactions?
The SN1 AND SN2 have few differences they are,- SN1 is unimolecular reaction(first order reaction), SN2 is bimolecular reaction(second order reaction).
- SN2 is stereospecific.
- SN2 depends on nuchleophile and substrate, SN1 depends only on substrate.
- SN2 occur in non-polar solvent.
- SN2 have transition state.
Why do high temperatures favor elimination reactions?
Elimination reactions require the breakage of more bonds than substitution reactions, and therefore require a higher input of energy. Increasing temperature will allow more molecules to reach the required activation energy threshold. Therefore, as temperatures is increased, elimination is favored.What is elimination reaction explain with example?
Elimination reactions are commonly known by the kind of atoms or groups of atoms leaving the molecule. The removal of a hydrogen atom and a halogen atom, for example, is known as dehydrohalogenation; when both leaving atoms are halogens, the reaction is known as dehalogenation.What happens in elimination reaction?
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism. In cases where the molecule is able to stabilize an anion but possesses a poor leaving group, a third type of reaction, E1CB, exists.What are the 4 main types of reactions?
Representation of four basic chemical reactions types: synthesis, decomposition, single replacement and double replacement.What is Saytzeff rule in organic chemistry?
Zaitsev's rule (or Saytzeff's rule, Saytzev's rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. More generally, Zaitsev's rule predicts that in an elimination reaction, the most substituted product will be the most stable, and therefore the most favored.What happens in an e1 reaction?
An E1 reaction involves the deprotonation of a hydrogen nearby (usually one carbon away, or the beta position) the carbocation resulting in the formation of an alkene product. Because it takes the electrons in the bond along with it, the carbon that was attached to it loses its electron, making it a carbocation.How many types of organic reactions are there?
fiveWhat does ch3oh do in a reaction?
Answer: CH3OH is a protic solvent. Because of hydrogen bonds, protic solvents decrease nucleophilicity. Therefore, CH3S- is a better nucleophile than CH3O- because smaller atoms have more concentrated electron density, so they hydrogen bond better. As a result, the reaction involving CH3O- is slower.Why is sn1 Unimolecular?
Because the slow step of the reaction involves only the substrate, the reaction is unimolecular. Because only the substrate is present in the transition state, the rate of the reaction depends only on its concentration, and not on the concentration of the nucleophile.Are sn1 reactions endothermic?
Ch 8 : SN1 mechanism. Since the first step of the reaction requires bond breaking (endothermic, heat in) to lose the leaving group, it will be the slow or rate determining step. Therefore, the general kinetic expression for an SN1 reaction is rate = k [ R-LG].What do e2 reactions favor?
From this diagram, it is evident that when substituents are arranged in a cis conformation torsional strain increases and stability decreases. Since E2 reactions generally favor the most stable product, the cis-alkene is typically not the favored product.Which alkyl halide reacts the fastest in an e2 reaction?
With 3rd degree alkyl halides, E2 reactions are favored over SN2 reactions. Tertiary Alkyl Halides undergo the fastest E2 reactions. The greater the alkyl substitution, the faster the reaction, since in the Transiton stage, a double bond is formed partially. A greater substituted alkene is lower in energy.Why is e2 reaction irreversible?
This E2 reaction is irreversible. The alkene products are not in equilibrium, so their relative stability does not control the amount of each product produced. Instead, the relative reaction rates control how much of each product is formed. This reaction is under kinetic control.What is required to promote an e2 reaction?
An E2 reaction has certain requirements to proceed:- Secondary and tertiary alkyl halides will proceed with E2 in the presence of a base (OH-, RO-, R2N-)
- Both leaving groups should be on the same plane, this allows the double bond to form in the reaction.
Which is least reactive in an e2 reaction?
Answer and Explanation: Compound A is least reactive towards E2 elimination reactions.Do e2 reactions change stereochemistry?
1 Answer. Ernest Z. The same atoms in the product are sp² hybridized, with bond angles of 120 °. This is a change in stereochemistry.What is e2 in chemistry?
E2 Definition. The E2 reaction - A Nucleophilic Elimination reaction in which the Rate Determining Step involves 2 components. -E2 reactions are bimolecular, with simultaneous bond-making and bond breaking steps.Is substitution or elimination faster?
Explanation: Elimination reactions compete with substitution reactions because both reaction mechanisms favour the same conditions: Alkyl halide and a nucleophile. The elimination will occur at a very fast rate and consume the alkyl halide before any substitution product is formed.Which is better substitution or elimination?
Substitution is best used when one (or both) of the equations is already solved for one of the variables. It also works well if one of the variables has a coefficient of 1. Elimination: Elimination is the method that I use almost every time. If you are not sure which method to use, I recommend that you use elimination.What is substitution and elimination reaction?
We will study three main types of reactions - addition, elimination and substitution. An addition reaction occurs when two or more reactants combine to form a single product. An elimination reaction occurs when a reactant is broken up into two products.How do you do substitution and elimination method?
How to solve linear systems with the elimination method- Rewrite the equations, if necessary, to make like variables line up underneath each other.
- Multiply the equations by constants to make one set of variables match coefficients.
- Add the two equations.
- Solve for the unknown variable that remains.